
Support
Preguntas frecuentes
Obtenga más información sobre nuestros productos, la escala ISQ y su uso, OsstellConnect, la tecnología Osstell y mucho más.

Questions about Smartpegs
Yes, it can be sterilised with a Prion cycle at 134 °C for 18 minutes.
Whether you are using a single-use or a reusable SmartPeg, it should always be installed using the SmartPeg Mount and tightened to approximately 4–6 Ncm – typically described as finger tight. This ensures proper seating and accurate ISQ measurements without risking damage to the implant or the SmartPeg.
Yes. You can mix single-use and reusable SmartPegs during the same treatment session. The only requirement is that you use the correct SmartPeg Type number for the specific implant or abutment you are measuring.
Whether the SmartPeg is single-use or reusable does not affect the measurement.
Yes, as long as its integrity and performance are maintained.
Osstell is also the only company offering both a sterile single-use and a reusable SmartPeg made of aluminium, supported by more than 1,500 published studies on the ISQ method.
With the Osstell original ISQ scale and SmartPegs, no conversion tables or correction factors are needed. The measured ISQ value is the true value, regardless of whether you use a single use or a reusable SmartPeg.
SmartPegs are supplied in different packaging formats depending on whether they are single-use or reusable:
Single-use SmartPegs
- Delivered sterile.
- Supplied in boxes of 5 pieces, in a single sterile barrier system with protective outer packaging.
- Intended for one treatment session only and must not be reused.
Reusable SmartPegs
- Delivered nonsterile in individual packaging.
- Must be cleaned, disinfected and sterilized before the first clinical use and after every subsequent use. (See quickguide at osstell/ifu).
SmartPeg Mount
- Delivered nonsterile with the instrument.
- Can be purchased separately, in package of 5.
- Must also be cleaned, disinfected and sterilized before use and between patients. (See quickguide at osstell/ifu).
Yes. The reusable and single use SmartPegs are designed so they can be easily distinguished visually:
- The single-use SmartPeg is supplied sterile, has an aluminium coloured (non coloured) magnet, and does not include any laser marked identifiers on the body.
- The reusable SmartPeg has a black magnet on top and carries laser marked Type and LOT numbers on the body for identification and traceability.

The reusable SmartPeg can be used for up to 20 reprocessing cycles, as long as its integrity and performance are maintained.
If inspection reveals wear, deformation, corrosion, damaged threads, or any loss of functional compatibility, the SmartPeg must be discarded, even if it has been processed fewer than 20 times.
The single-use SmartPeg is intended for one treatment session only and must not be reprocessed.
The reusable SmartPeg is engineered with special protective surface coating – one on the SmartPeg body and another black protective coating on the magnet. These coatings provide resistance to moisture, cleaning chemicals, wear, and corrosion, ensuring that the SmartPeg maintains its performance throughout the validated number of reprocessing cycles.
The reusable SmartPeg can easily be distinguished from the single use version: it has a black magnet, and its LOT and Type number are laser marked on the body for clear identification.
Prior to the first clinical use, and after each use the reusable products must be cleaned, disinfected and sterilized according to the instructions in the quickguide that is found here: Osstell/ifu
You can record ISQ at abutment level in OsstellConnect, but it is important to follow the correct clinical measurement procedure to obtain accurate values.
How to measure ISQ at abutment level
- Perform an implant level measurement first, before placing the abutment.
- Measure again at abutment level during the same session, using a SmartPeg made for abutment level measuring.
- Determine the offset by noting the difference between implant level and abutment level ISQ. This offset remains stable throughout healing and should be used when interpreting follow-up measurements.
- For all subsequent visits, measure on the abutment and apply the same offset to understand the true implant ISQ.
How to record the measurement in OsstellConnect:
- Go to the patient’s record, select the patient and the implant in the tooth chart. Click ‘Add abutment’, choose the abutment, and save.
- Select ‘Measure stability’ and choose ‘Abutment’ under Measurement level. Measure on the abutment as you would at implant level and save.
- To review the stability trend, open the Stability tab and toggle ‘Show abutment’. You can also enable ‘Show details’ to view buccal–lingual and mesial–distal ISQ values.
For a more detailed explanation and clinical guidance, see our blog post here.
No. A torque device should not be used to tighten the SmartPeg.
SmartPegs should always be mounted using the SmartPeg Mount and tightened to approximately 4–6 Ncm, which is typically described as finger tight.
This ensures the SmartPeg is seated correctly without risking damage to the implant connection.
ISQ should always be measured in two directions, because implant stability can vary depending on the amount of surrounding bone in different directions. Measuring from two angles ensures that both the most and the least stable directions are captured.
To do this, take one measurement in the buccal-lingual direction and one in the mesial-distal direction. These two ISQ values represent the different stability directions. Sometimes the values are identical; sometimes they differ.
If the two measurements differ, always rely on the lowest ISQ value, as it reflects the weakest direction and therefore provides the most clinically relevant information about implant stability.
Osstell measures implant stability using Resonance Frequency Analysis (RFA). The small SmartPeg that you attach to the implant has a tiny magnet on top. When the probe sends magnetic pulses, the SmartPeg starts to vibrate – similar to a very small tuning fork.
How it works:
- The probe sends magnetic pulses
These pulses make the SmartPeg vibrate without touching it. - The vibration changes depending on how stable the implant is
– A stable implant (good bone support) makes the SmartPeg vibrate faster.
– A less stable implant makes it vibrate slower. - The device converts this vibration into an ISQ value
– Higher stability → higher frequency → higher ISQ
– Lower stability → lower frequency → lower ISQ
It takes less than a second, and the ISQ value is shown immediately.
Yes. The ISQ value can be affected if the SmartPeg is not mounted correctly.
For accurate measurements, the SmartPeg should be attached finger tight using the SmartPeg Mount, which corresponds to about 4–6 Ncm.
At this level, the SmartPeg has firm, stable contact with the implant, and the resulting ISQ value is consistent – typically within ±2 ISQ units.
- If the SmartPeg is not fully seated:
The contact becomes unstable, and the ISQ value can appear artificially low, or you don’t receive any value. - If too much force is applied:
Overtightening does not improve accuracy. It may damage the SmartPeg threads.
– A reusable SmartPeg may be damaged so it cannot be used again.
– A single use or reusable SmartPeg may have its threads deformed, which can lead to inaccurate measurements during that session or, in worst case a broken SmartPeg.
In short: finger tight (4–5 Ncm) is enough to ensure stable contact and reliable ISQ readings.
If no ISQ value appears, it usually means the SmartPeg’s vibration is not being detected clearly by the instrument. Several factors can cause this, and most are easy to resolve:
- Distance to instrument is too great
The instrument should normally be held 2–4 mm from the magnet on top of the SmartPeg. Do not touch the magnet. If no value appears, move the instrument closer until the signal is detected. - Angle needs adjustment
The magnetic field is directional, so the orientation of the instrument matters. Measurements should be taken at approximately a 45° angle relative to the top of the SmartPeg. - Ensure you have the correct SmartPeg Type for the specific implant
Use this link to find the correct SmartPeg Type: Find my SmartPeg - Soft tissue damping
Surrounding tissue can sometimes dampen the vibration more than expected. Holding the instrument a bit closer usually compensates for this. - The implant platform is not clean
Ensure the implant platform is clean and free of debris, so the SmartPeg seats properly and transmits its vibration. - The SmartPeg is worn or damaged
• If you are using a reusable SmartPeg, make sure it has been inspected before use and that it is within 20 reprocessing cycles. A reusable SmartPeg that is worn out, bent, or has damaged threads will not vibrate correctly — and the instrument may not be able to pick up a reading.
• A single‑use SmartPeg is intended for one treatment session only.
• Ensure the SmartPeg is properly seated on the implant and that it is not in contact with surrounding tissue, as this can prevent accurate signal detection and may result in no ISQ value or an incorrect one. - Electromagnetic interference
Strong electromagnetic noise sources nearby may disturb the measurement. (See the FAQ on “Electromagnetic noise” for details.)
The following products may be autoclaved (over-pressurized steam up to 135 deg Celsius):
- SmartPeg Mount
- Reusable SmartPeg
- Probe (IDx)
Please note that single-use SmartPegs are disposable and cannot be autoclaved!
You can check the SmartPeg Reference Guide by clicking here
Questions about the Osstell Beacon
You can record ISQ at abutment level in OsstellConnect, but it is important to follow the correct clinical measurement procedure to obtain accurate values.
How to measure ISQ at abutment level
- Perform an implant level measurement first, before placing the abutment.
- Measure again at abutment level during the same session, using a SmartPeg made for abutment level measuring.
- Determine the offset by noting the difference between implant level and abutment level ISQ. This offset remains stable throughout healing and should be used when interpreting follow-up measurements.
- For all subsequent visits, measure on the abutment and apply the same offset to understand the true implant ISQ.
How to record the measurement in OsstellConnect:
- Go to the patient’s record, select the patient and the implant in the tooth chart. Click ‘Add abutment’, choose the abutment, and save.
- Select ‘Measure stability’ and choose ‘Abutment’ under Measurement level. Measure on the abutment as you would at implant level and save.
- To review the stability trend, open the Stability tab and toggle ‘Show abutment’. You can also enable ‘Show details’ to view buccal–lingual and mesial–distal ISQ values.
For a more detailed explanation and clinical guidance, see our blog post here.
Although it is possible to use the Osstell Beacon directly, depending on the status of the built-in rechargeable battery, it is recommended to charge the battery for at least 3 hours before using the Osstell Beacon instrument.
The Osstell Beacon is activated by a built-in motion sensor. As soon as the motion sensor detects movement, the Osstell Beacon will start-up and shortly show battery status and then be ready for measurement in the BL (Buccal – Lingual) direction, which is indicated in the upper display.
The Osstell Beacon will start to measure, indicated by an audible sound, when near an Osstell SmartPeg or Test peg due to a built-in magnetic detector that senses the magnet on the top of the Osstell SmartPeg/TestPeg. Measured data will be shown in the upper display combined with a colored light indication below the tip.
The red, yellow and green color indicates if the measured ISQ value corresponds to a low, medium or high implant stability according to the ISQ scale guidance, click here for more information.
After 60 seconds of no motion, the Osstell Beacon will turn off automatically.
1. Activate the Osstell Beacon by picking it up. The instrument will start-up and after showing the battery status, the instrument will be ready for measurement in the BL (Buccal – Lingual) direction, which is prompted in the upper display as well as optimal angle towards the Smartpeg.
2. Place a barrier sleeve over the Osstell beacon instrument.
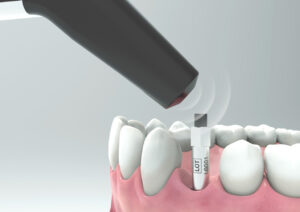
3. Place the SmartPeg into the SmartPeg Mount. The SmartPeg is magnetic, and the SmartPeg Mount will hold the SmartPeg. See fig 2. Attach the SmartPeg to the implant or abutment by screwing the SmartPeg Mount using finger force of approximately 4-6 Ncm. Do not over-tighten, to avoid damaging the SmartPeg threads.
4. Hold the instrument tip close (2-4mm) to the top of the SmartPeg without touching it. Sometimes, you will need to go as close as 1 mm. An audible sound indicates when measurement has started, and measured data will be shown in the upper display combined with a colored light indication below the tip.
5. Bring the instrument out of the mouth to clearly read the ISQ value and the colored indication. The measured ISQ values will be displayed in the upper display for a couple of seconds and then switch to be ready for measurement in mesial-distal direction.
6. Repeat step 4 and the sequence starts over and the Osstell Beacon is ready for measurement in the BL (Buccal- Lingual) direction again.
7. When all measurements are performed, remove the SmartPeg by using the SmartPeg Mount. The instrument is automatically turned off after 60 seconds of inactivity.
Algunas posibles explicaciones son:
SmartPeg reutilizado: Los SmartPegs son desechables y solo deben utilizarse para una o varias mediciones en una sesión de tratamiento, para un solo paciente. La reutilización repetida puede dar lugar a lecturas falsas debido al desgaste de las roscas blandas de aluminio del SmartPeg.
Tipo de SmartPeg incorrecto seleccionado para el implante: Consulte la lista de referencia de SmartPeg, staging.osstell.com/smartpegguide
Hueso o tejido blando entre SmartPeg e implante: Asegúrese de limpiar la conexión protésica del implante antes de colocar el SmartPeg.
Interferencias electromagnéticas: Retire la fuente de las interferencias electromagnéticas.
La sonda está demasiado lejos del SmartPeg: Sostenga la sonda a 2-4 mm del SmartPeg.
Although it is possible to use the Osstell Beacon directly, depending on the status of the built-in rechargeable battery, it is recommended to charge the battery for at least 3 hours before using the Osstell Beacon instrument.
The Osstell Beacon is activated by a built-in motion sensor. As soon as the motion sensor detects movement, the Osstell Beacon will start-up and shortly show battery status and then be ready for measurement in the BL (Buccal – Lingual) direction, which is indicated in the upper display.
The Osstell Beacon will start to measure, indicated by an audible sound, when near an Osstell SmartPeg or Test peg due to a built-in magnetic detector that senses the magnet on the top of the Osstell SmartPeg/TestPeg. Measured data will be shown in the upper display combined with a colored light indication below the tip.
The red, yellow and green color indicates if the measured ISQ value corresponds to a low, medium or high implant stability according to the ISQ scale guidance, click here for more information.
1. Activate the Osstell Beacon by picking it up. The instrument will start-up and after showing the battery status, the instrument will be ready for measurement in the BL (Buccal – Lingual) direction, which is prompted in the upper display as well as optimal angle towards the Smartpeg.
2. Place a barrier sleeve over the Osstell beacon instrument.
3. Place the SmartPeg into the SmartPeg Mount. The SmartPeg is magnetic, and the SmartPeg Mount will hold the SmartPeg. See fig 2. Attach the SmartPeg to the implant or abutment by screwing the SmartPeg Mount using finger force of approximately 4-6 Ncm. Do not over-tighten, to avoid damaging the SmartPeg threads.
4. Hold the instrument tip close (2-4mm) to the top of the SmartPeg without touching it. Sometimes, you will need to go as close as 1 mm. An audible sound indicates when measurement has started, and measured data will be shown in the upper display combined with a colored light indication below the tip.
5. Bring the instrument out of the mouth to clearly read the ISQ value and the colored indication. The measured ISQ values will be displayed in the upper display for a couple of seconds and then switch to be ready for measurement in mesial-distal direction.
6. Repeat step 4 and the sequence starts over and the Osstell Beacon is ready for measurement in the BL (Buccal- Lingual) direction again.
7. When all measurements are performed, remove the SmartPeg by using the SmartPeg Mount. The instrument is automatically turned off after 60 seconds of inactivity.
Possible explanations include:
Re-Used SmartPeg: The SmartPegs are disposable and should only be used for one or multiple measurement at one treatment session, for a single patient only. Repeated re-use may result in false readings due to wear and tear of the soft aluminum SmartPeg threads.
Wrong SmartPeg type selected for the implant: See SmartPeg reference list, staging.osstell.com/smartpegguide
Bone or soft tissue in between SmartPeg and Implant: Make sure to clean the Implant prosthetic connection before attaching the SmartPeg.
Electromagnetic interference: Remove the source of electromagnetic interference.
Probe is held too far away from the SmartPeg: Hold the probe 2-4 mm away from the SmartPeg.
You can have your Osstell Beacon online connected to OsstellConnect for data collection, but it is not a requirement. OsstellConnect is an online tool for statistical analysis and valuable insights based on your data. Before you can start using data connection to OsstellConnect you have to register your Osstell Beacon. The serial number can be found on the back of the instrument. For registration assistance and features of data connection to OsstellConnect, please visit osstellconnect.com/beacon
2 years
Before each use, moisten a gauze or soft cloth with an acceptable (see list below) surface disinfectant and wipe the whole Osstell Beacon instrument. Note! Do not autoclave the Osstell Beacon instrument. Routinely check the surface of the instrument tip and overall surface for possible cracks and resins.
Acceptable Cleaners:
o Isopropyl alcohol
o Ethyl alcohol-based cleaners
Do not use:
o Strong alkali detergent of any type, including hand soaps and dish soaps Bleach based cleaners
o Hydrogen Peroxide based cleaners Abrasive Cleansers
o Acetone or hydrocarbon based cleaners MEK (Methyl Ethyl Ketone)
o Birex Gluteraldehyde
o Quaternary Ammonium Chloride salt-based cleaners
The Osstell Beacon is delivered from factory in a “transport” mode, where the built-in motion sensor is deactivated. To deactivate the transport mode and start charging, connect the small USB connector of the Osstell USB cable to wide end of the instrument. Connect the large USB connector to a standard USB outlet of a PC, laptop or charger.
You can have your Osstell Beacon online connected to OsstellConnect for data collection, but it is not a requirement. OsstellConnect is an online tool for statistical analysis and valuable insights based on your data. Before you can start using data connection to OsstellConnect you have to register your Osstell Beacon. The serial number can be found on the back of the instrument. For registration assistance and features of data connection to OsstellConnect, please visit osstellconnect.com/beacon
2 years
Before each use, moisten a gauze or soft cloth with an acceptable (see list below) surface disinfectant and wipe the whole Osstell Beacon instrument. Note! Do not autoclave the Osstell Beacon instrument. Routinely check the surface of the instrument tip and overall surface for possible cracks and resins.
Acceptable Cleaners:
o Isopropyl alcohol
o Ethyl alcohol-based cleaners
Do not use:
o Strong alkali detergent of any type, including hand soaps and dish soaps
o Bleach based cleaners
o Hydrogen Peroxide based cleaners
o Abrasive Cleansers
o Acetone or hydrocarbon based cleaners
o MEK (Methyl Ethyl Ketone)
o Birex
o Gluteraldehyde
o Quaternary Ammonium Chloride salt-based cleaners
The Osstell Beacon is delivered from factory in a “transport” mode, where the built-in motion sensor is deactivated. To deactivate the transport mode and start charging, connect the small USB connector of the Osstell USB cable to wide end of the instrument. Connect the large USB connector to a standard USB outlet of a PC, laptop or charger.
Charge the Beacon for at least 30 min in room temperature, then try again. If the error remains, try resetting the device by:
1. Identify the Beacon reset pin on the picture below.
![]()
2. With a paper clip, do a gentle press for 1 second on the reset button (Note! You may have to tilt the pin a little downwards).
If you see a “PadLock” symbol in the upper display, press one more time on the reset button.
If the Beacon still doesn’t start or the display is showing a wrench and a manual, then please contact your local sales representative or distributor or support@staging.osstell.com.
In general, Electromagnetic noise, or should be taken as an indication that the environment contains an alternating magnetic field of some magnitude. However there could be sudden bursts of electromagnetic noise from different electronic equipment, which could sometimes coincide with a measurement. TV-sets are among those devices that create a lot of noise.
If the probe is close to the instrument (a few centimeters), the noise from the instrument itself will cause the warning. If the warning is only visible when the probe is held close to a SmartPeg, it can be due to:
– If the implant is really rigid, it makes the signal from the SmartPeg very strong, which can cause the warning. Try to distance the probe from the SmartPeg a millimeter or two. Clinically this should happen very seldom, but on a plastic jaw model for instance, it can be necessary to hold the probe several millimeters from the SmartPeg, due to the lack of dampening soft tissue.
– If the SmartPeg is not mounted properly, it can cause the warning. If this has happened the SmartPeg threads are probably damaged, and it has to be exchanged.
There are currently thousands of articles where Osstell was used in the study, published in scientific papers. These abstracts could be found in our searchable database under “Resources/Scientific database“.
ISQ should always be measured in two directions, because implant stability can vary depending on the amount of surrounding bone in different directions. Measuring from two angles ensures that both the most and the least stable directions are captured.
To do this, take one measurement in the buccal-lingual direction and one in the mesial-distal direction. These two ISQ values represent the different stability directions. Sometimes the values are identical; sometimes they differ.
If the two measurements differ, always rely on the lowest ISQ value, as it reflects the weakest direction and therefore provides the most clinically relevant information about implant stability.
Osstell measures implant stability using Resonance Frequency Analysis (RFA). The small SmartPeg that you attach to the implant has a tiny magnet on top. When the probe sends magnetic pulses, the SmartPeg starts to vibrate – similar to a very small tuning fork.
How it works:
- The probe sends magnetic pulses
These pulses make the SmartPeg vibrate without touching it. - The vibration changes depending on how stable the implant is
– A stable implant (good bone support) makes the SmartPeg vibrate faster.
– A less stable implant makes it vibrate slower. - The device converts this vibration into an ISQ value
– Higher stability → higher frequency → higher ISQ
– Lower stability → lower frequency → lower ISQ
It takes less than a second, and the ISQ value is shown immediately.
If no ISQ value appears, it usually means the SmartPeg’s vibration is not being detected clearly by the instrument. Several factors can cause this, and most are easy to resolve:
- Distance to instrument is too great
The instrument should normally be held 2–4 mm from the magnet on top of the SmartPeg. Do not touch the magnet. If no value appears, move the instrument closer until the signal is detected. - Angle needs adjustment
The magnetic field is directional, so the orientation of the instrument matters. Measurements should be taken at approximately a 45° angle relative to the top of the SmartPeg. - Ensure you have the correct SmartPeg Type for the specific implant
Use this link to find the correct SmartPeg Type: Find my SmartPeg - Soft tissue damping
Surrounding tissue can sometimes dampen the vibration more than expected. Holding the instrument a bit closer usually compensates for this. - The implant platform is not clean
Ensure the implant platform is clean and free of debris, so the SmartPeg seats properly and transmits its vibration. - The SmartPeg is worn or damaged
• If you are using a reusable SmartPeg, make sure it has been inspected before use and that it is within 20 reprocessing cycles. A reusable SmartPeg that is worn out, bent, or has damaged threads will not vibrate correctly — and the instrument may not be able to pick up a reading.
• A single‑use SmartPeg is intended for one treatment session only.
• Ensure the SmartPeg is properly seated on the implant and that it is not in contact with surrounding tissue, as this can prevent accurate signal detection and may result in no ISQ value or an incorrect one. - Electromagnetic interference
Strong electromagnetic noise sources nearby may disturb the measurement. (See the FAQ on “Electromagnetic noise” for details.)

No. The Osstell measurement does not affect the implant. The instrument only makes the SmartPeg vibrate. The SmartPeg’s magnet is activated by very low energy magnetic pulses generated by the instrument. The technique is objective, gentle, and highly repeatable

Preguntas sobre Osstell Classic
1 año.
Antes de utilizar el instrumento en un paciente, coloque una funda protectora sobre él. La funda protectora ayuda a evitar la contaminación cruzada y a impedir que el material dental compuesto se adhiera a la superficie de la punta y del cuerpo del instrumento y que las soluciones de limpieza produzcan manchas y degradación.
Nota
- Las fundas protectoras son para uso en un solo paciente.
- Deseche las fundas protectoras usadas en la basura normal después de cada paciente.
- No deje las fundas protectoras en el instrumento durante periodos prolongados.
- Consulte las fundas protectoras recomendadas a continuación.
- TIDIShield, n.° art.: 20808, n.° art.: 20987. tidiproducts.com
- PremiumPlus: Funda del sensor de rayos X 183-2, tamaño 2
- Después de usarlo en cada paciente, el instrumento debe limpiarse con los productos adecuados.
- Debe realizarse una primera medición en el momento de la colocación del implante para obtener un valor de referencia para futuras mediciones a lo largo del proceso de cicatrización. Antes de la restauración definitiva, se realiza otra medición que permite observar la evolución de la estabilidad del implante.
- Se recomienda medir tanto en dirección bucolingual como mesiodistal para encontrar la estabilidad más baja. Por lo tanto, el instrumento solicita al usuario que realice la medición en ambas direcciones.
- Haga clic aquí para ver un vídeo sobre cómo medir.
Antes de cada uso, humedezca una gasa o un paño suave con un limpiador de superficies recomendado (véase la lista más abajo) y limpie todo el instrumento.
Limpiadores aceptables:
- Alcohol isopropílico al 70%
- Detergentes enzimáticos con pH neutro que hagan poca espuma, por ejemplo:
- Toallitas desinfectantes BePro, REF. 19500102, www.wh.com (se pueden pedir a W&H Sterilization)
- Medizime LF
- Enzol
No use:
- Limpiadores o desinfectantes con base ácida o fenólica
- Cualquier tipo de detergente alcalino fuerte, incluidos los jabones de manos y lavavajillas
- Limpiadores con base de lejía
- Limpiadores con base de peróxido de hidrógeno
- Limpiadores abrasivos
- Limpiadores con base de acetona o de hidrocarburo
- MEK (metiletilcetona)
- Birex
- Gluteraldehído
- Limpiadores con base de sales de cloruro de amonio cuaternario
El instrumento no requiere mantenimiento regular. En caso de avería del instrumento, póngase en contacto con el representante de ventas o distribuidor local para obtener más instrucciones.
You can record ISQ at abutment level in OsstellConnect, but it is important to follow the correct clinical measurement procedure to obtain accurate values.
How to measure ISQ at abutment level
- Perform an implant level measurement first, before placing the abutment.
- Measure again at abutment level during the same session, using a SmartPeg made for abutment level measuring.
- Determine the offset by noting the difference between implant level and abutment level ISQ. This offset remains stable throughout healing and should be used when interpreting follow-up measurements.
- For all subsequent visits, measure on the abutment and apply the same offset to understand the true implant ISQ.
How to record the measurement in OsstellConnect:
- Go to the patient’s record, select the patient and the implant in the tooth chart. Click ‘Add abutment’, choose the abutment, and save.
- Select ‘Measure stability’ and choose ‘Abutment’ under Measurement level. Measure on the abutment as you would at implant level and save.
- To review the stability trend, open the Stability tab and toggle ‘Show abutment’. You can also enable ‘Show details’ to view buccal–lingual and mesial–distal ISQ values.
For a more detailed explanation and clinical guidance, see our blog post here.
Algunas posibles explicaciones son:
SmartPeg reutilizado: Los SmartPegs son desechables y solo deben utilizarse para una o varias mediciones en una sesión de tratamiento, para un solo paciente. La reutilización repetida puede dar lugar a lecturas falsas debido al desgaste de las roscas blandas de aluminio del SmartPeg.
Tipo de SmartPeg incorrecto seleccionado para el implante: Consulte la lista de referencia de SmartPeg, staging.osstell.com/smartpegguide
Hueso o tejido blando entre SmartPeg e implante: Asegúrese de limpiar la conexión protésica del implante antes de colocar el SmartPeg.
Interferencias electromagnéticas: Retire la fuente de las interferencias electromagnéticas.
La sonda está demasiado lejos del SmartPeg: Sostenga la sonda a 2-4 mm del SmartPeg.
In general, Electromagnetic noise, or should be taken as an indication that the environment contains an alternating magnetic field of some magnitude. However there could be sudden bursts of electromagnetic noise from different electronic equipment, which could sometimes coincide with a measurement. TV-sets are among those devices that create a lot of noise.
If the probe is close to the instrument (a few centimeters), the noise from the instrument itself will cause the warning. If the warning is only visible when the probe is held close to a SmartPeg, it can be due to:
– If the implant is really rigid, it makes the signal from the SmartPeg very strong, which can cause the warning. Try to distance the probe from the SmartPeg a millimeter or two. Clinically this should happen very seldom, but on a plastic jaw model for instance, it can be necessary to hold the probe several millimeters from the SmartPeg, due to the lack of dampening soft tissue.
– If the SmartPeg is not mounted properly, it can cause the warning. If this has happened the SmartPeg threads are probably damaged, and it has to be exchanged.
There are currently thousands of articles where Osstell was used in the study, published in scientific papers. These abstracts could be found in our searchable database under “Resources/Scientific database“.
ISQ should always be measured in two directions, because implant stability can vary depending on the amount of surrounding bone in different directions. Measuring from two angles ensures that both the most and the least stable directions are captured.
To do this, take one measurement in the buccal-lingual direction and one in the mesial-distal direction. These two ISQ values represent the different stability directions. Sometimes the values are identical; sometimes they differ.
If the two measurements differ, always rely on the lowest ISQ value, as it reflects the weakest direction and therefore provides the most clinically relevant information about implant stability.
Osstell measures implant stability using Resonance Frequency Analysis (RFA). The small SmartPeg that you attach to the implant has a tiny magnet on top. When the probe sends magnetic pulses, the SmartPeg starts to vibrate – similar to a very small tuning fork.
How it works:
- The probe sends magnetic pulses
These pulses make the SmartPeg vibrate without touching it. - The vibration changes depending on how stable the implant is
– A stable implant (good bone support) makes the SmartPeg vibrate faster.
– A less stable implant makes it vibrate slower. - The device converts this vibration into an ISQ value
– Higher stability → higher frequency → higher ISQ
– Lower stability → lower frequency → lower ISQ
It takes less than a second, and the ISQ value is shown immediately.
If no ISQ value appears, it usually means the SmartPeg’s vibration is not being detected clearly by the instrument. Several factors can cause this, and most are easy to resolve:
- Distance to instrument is too great
The instrument should normally be held 2–4 mm from the magnet on top of the SmartPeg. Do not touch the magnet. If no value appears, move the instrument closer until the signal is detected. - Angle needs adjustment
The magnetic field is directional, so the orientation of the instrument matters. Measurements should be taken at approximately a 45° angle relative to the top of the SmartPeg. - Ensure you have the correct SmartPeg Type for the specific implant
Use this link to find the correct SmartPeg Type: Find my SmartPeg - Soft tissue damping
Surrounding tissue can sometimes dampen the vibration more than expected. Holding the instrument a bit closer usually compensates for this. - The implant platform is not clean
Ensure the implant platform is clean and free of debris, so the SmartPeg seats properly and transmits its vibration. - The SmartPeg is worn or damaged
• If you are using a reusable SmartPeg, make sure it has been inspected before use and that it is within 20 reprocessing cycles. A reusable SmartPeg that is worn out, bent, or has damaged threads will not vibrate correctly — and the instrument may not be able to pick up a reading.
• A single‑use SmartPeg is intended for one treatment session only.
• Ensure the SmartPeg is properly seated on the implant and that it is not in contact with surrounding tissue, as this can prevent accurate signal detection and may result in no ISQ value or an incorrect one. - Electromagnetic interference
Strong electromagnetic noise sources nearby may disturb the measurement. (See the FAQ on “Electromagnetic noise” for details.)

No. The Osstell measurement does not affect the implant. The instrument only makes the SmartPeg vibrate. The SmartPeg’s magnet is activated by very low energy magnetic pulses generated by the instrument. The technique is objective, gentle, and highly repeatable
Comprender la osteointegración en odontología
Questions about OsstellConnect
You can record ISQ at abutment level in OsstellConnect, but it is important to follow the correct clinical measurement procedure to obtain accurate values.
How to measure ISQ at abutment level
- Perform an implant level measurement first, before placing the abutment.
- Measure again at abutment level during the same session, using a SmartPeg made for abutment level measuring.
- Determine the offset by noting the difference between implant level and abutment level ISQ. This offset remains stable throughout healing and should be used when interpreting follow-up measurements.
- For all subsequent visits, measure on the abutment and apply the same offset to understand the true implant ISQ.
How to record the measurement in OsstellConnect:
- Go to the patient’s record, select the patient and the implant in the tooth chart. Click ‘Add abutment’, choose the abutment, and save.
- Select ‘Measure stability’ and choose ‘Abutment’ under Measurement level. Measure on the abutment as you would at implant level and save.
- To review the stability trend, open the Stability tab and toggle ‘Show abutment’. You can also enable ‘Show details’ to view buccal–lingual and mesial–distal ISQ values.
For a more detailed explanation and clinical guidance, see our blog post here.
• To document an implant failure, go to your patients records. Select the patient, then select the tooth. Click ‘Mark as failed’. Choose the type of failure (early or late failure), fill in the failure date. Choose the reason for failure. Click ‘Mark as failed’.
• You can access the history of the implant failure by navigating to the history tab. Click the tab ‘Failed’ to see all information related to the treatment and the failure.
• A history symbol is displayed on the tooth with a failed implant.
The data in your Osstell IDx is automatically uploaded to your OsstellConnect account, if it is connected to the internet. To connect your device to the internet, please follow the instructions here.
Please note that you can also create backups, for example on a USB stick. Please follow the instructions here.
Before starting, make sure that you have already installed the Beacon Gateway. If you have not installed it yet, please navigate to your clinic in your account, click the instruments tab and download the gateway. Install it.
• Plug in the Osstell Key to a USB port on your computer.
• Take your instrument in hand. It is now connecting to OsstellConnect and you should see the following symbol animating. Please note that it can take a few seconds.
• When connected, the symbol stops animating.
• Go to your patient records.
—— If it is the first measurement, create a patient. Fill in patient data and save. Click New Treatment and fill in treatment information. Click Add implant, fill in implant data and save. Click measure stability.
—— If it is a follow up measurement, select the patient and then the tooth on the teeth chart. Click measure stability.
• Measure in buccal lingual direction. The ISQ value is now showing in OsstellConnect.
• Measure in mesial distal direction. The ISQ value is now showing in OsstellConnect.
• Fill in the information about the treatment and save.
Navigate to your clinic in your account. Select the instrument tab. Click on Download firmware.
Download instructions:
• Wait for the download to complete (could take a while depending on your connection)
• Copy the .m2f binary file to a USB memory stick
• Insert the memory stick in your IDx while it is running
• Follow instructions on the IDx display
Note: you cannot open or view the .m2f file
You can also check this guide
Yes, simply navigate to your patient records. Select the patient. Select the implant. Go to the stability tab. Click on the edit icon by the measurement you would like to delete. Click delete. Please note that the measurement will be permanently removed from the patient chart.
Yes, simply navigate to your patient records. Select the patient you would like to delete. Click delete in the upper right corner. Confirm that you want to delete the patient and all data. Please note that it will be permanently removed from your account.
Yes, you can export all your data to an Excel file for further analysis. Simply go to your patients records and click CSV export. The file will appear in your downloads.
Yes, simply navigate to your patient records. Select the patient. Select the implant you want to edit on the tooth chart. Select the tab ‘Implant & abutment’. Click on the pen icon to edit. Edit implant data. Save.
Simply navigate to your patient records. Select the patient. Click ‘edit’ on the top right corner. Edit the data and save.
You can find the serial number on the back of your instrument.
Osstell Beacon
• To find your registration code, please make sure that you have already installed the Beacon Gateway. If you have not installed it yet, please navigate to your clinic in your account, click the instruments tab and download the gateway. Install it.
• Make sure that you have inserted the Osstell Key to your computer.
• Take your instrument in hand. It is now connecting to OsstellConnect and you should see a symbol animating. Please note that it can take a few seconds.
• When connected, the symbol stops animating.
• The registration code is now displayed on the lower screen of your instrument.
Osstell IDx
• When turning on your Osstell IDx for the first time, you will be prompted to select language, time zone, date and time.
• After that, you will need to set up a 4-digit pin code and a backup password. The backup password is needed when restoring to a backup, so please note it down and save it in a safe place.
• Your registration code is now displayed on your Osstell IDx.
ISQ should always be measured in two directions, because implant stability can vary depending on the amount of surrounding bone in different directions. Measuring from two angles ensures that both the most and the least stable directions are captured.
To do this, take one measurement in the buccal-lingual direction and one in the mesial-distal direction. These two ISQ values represent the different stability directions. Sometimes the values are identical; sometimes they differ.
If the two measurements differ, always rely on the lowest ISQ value, as it reflects the weakest direction and therefore provides the most clinically relevant information about implant stability.

Questions about the Osstell IDx
You can record ISQ at abutment level in OsstellConnect, but it is important to follow the correct clinical measurement procedure to obtain accurate values.
How to measure ISQ at abutment level
- Perform an implant level measurement first, before placing the abutment.
- Measure again at abutment level during the same session, using a SmartPeg made for abutment level measuring.
- Determine the offset by noting the difference between implant level and abutment level ISQ. This offset remains stable throughout healing and should be used when interpreting follow-up measurements.
- For all subsequent visits, measure on the abutment and apply the same offset to understand the true implant ISQ.
How to record the measurement in OsstellConnect:
- Go to the patient’s record, select the patient and the implant in the tooth chart. Click ‘Add abutment’, choose the abutment, and save.
- Select ‘Measure stability’ and choose ‘Abutment’ under Measurement level. Measure on the abutment as you would at implant level and save.
- To review the stability trend, open the Stability tab and toggle ‘Show abutment’. You can also enable ‘Show details’ to view buccal–lingual and mesial–distal ISQ values.
For a more detailed explanation and clinical guidance, see our blog post here.
Two years.
The IDx consumes power when it is turned off, so if the battery is totally drained and down at 0% the settings for date and time will be lost. If the battery is drained the IDx will ask you to set date and time again at start-up. If you connect the IDx to the internet, the date and time will be set automatically.
Simply register your IDx by following the instructions in the device or by following this link.
ISQ should always be measured in two directions, because implant stability can vary depending on the amount of surrounding bone in different directions. Measuring from two angles ensures that both the most and the least stable directions are captured.
To do this, take one measurement in the buccal-lingual direction and one in the mesial-distal direction. These two ISQ values represent the different stability directions. Sometimes the values are identical; sometimes they differ.
If the two measurements differ, always rely on the lowest ISQ value, as it reflects the weakest direction and therefore provides the most clinically relevant information about implant stability.
No, for the moment is it not possible to connect to other patient data system.
For normal use, the batteries last for at least one day but in most cases longer. It takes about 2
hrs to charge the IDx from a fully discharged state.
Simply choose a tooth position on the touch screen and start measuring.
No. The Osstell measurement does not affect the implant. The instrument only makes the SmartPeg vibrate. The SmartPeg’s magnet is activated by very low energy magnetic pulses generated by the instrument. The technique is objective, gentle, and highly repeatable
Perform measurements, press End patient, and then save as Add new patient.
Simply select the pen symbol in the patient list to edit.
The patient data can be saved either in the instrument or on Osstell Connect.
Values can only be removed before saving clicking on the trash bin symbol. After saving the data cannot be modified.
Yes, OsstellConnect is accessible via iPhone, iPad, Mac or PC.
Have you registered your device? You can only use the Osstell IDx for a limited time without
registration.
The following products may be autoclaved (over-pressurized steam up to 135 deg Celsius):
- SmartPeg Mount
- Reusable SmartPeg
- Probe (IDx)
Please note that single-use SmartPegs are disposable and cannot be autoclaved!
No, you can also register your IDx at osstellconnect.com. All you need is the serial number.
To enable automatic backup of your Osstell IDx, you will be asked to create a backup code made up of 8 characters. The purpose of the backup is to reset the unit if you for some reason remove patients by mistake or lose your instrument. The code is created during the registration process and you only have to enter it once. To make sure you can access your backup at a later date, it is VERY important that you write this code down and keep it in a safe place.
Backup can also be done via USB without a code.
Just connect the instrument via wifi and follow the instructions on the screen. A confirmation
email with login details will be sent to your email address.
Questions about the Osstell ISQ
ISQ should always be measured in two directions, because implant stability can vary depending on the amount of surrounding bone in different directions. Measuring from two angles ensures that both the most and the least stable directions are captured.
To do this, take one measurement in the buccal-lingual direction and one in the mesial-distal direction. These two ISQ values represent the different stability directions. Sometimes the values are identical; sometimes they differ.
If the two measurements differ, always rely on the lowest ISQ value, as it reflects the weakest direction and therefore provides the most clinically relevant information about implant stability.
Questions about Osstell and the ISQ scale
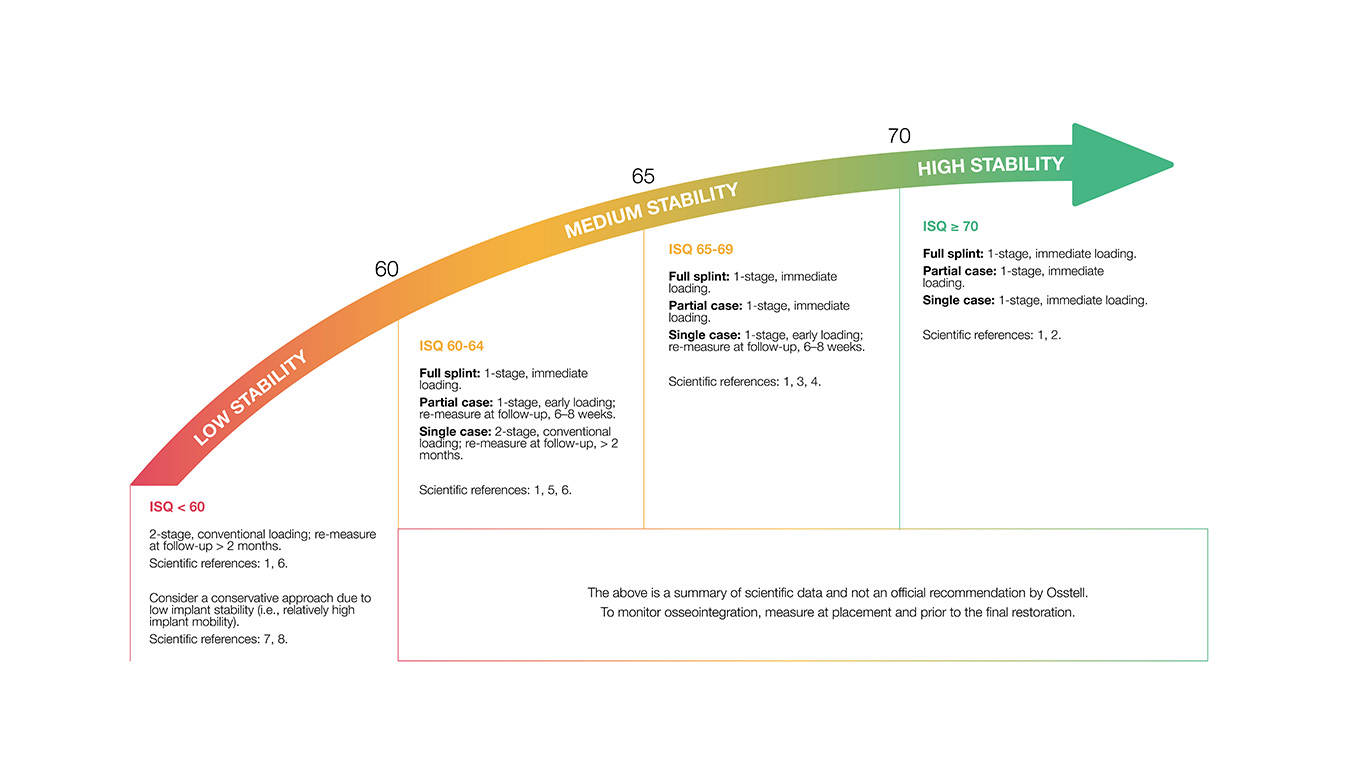
ISQ means Implant Stability Quotient and derives from Herz since the technique we use is Resonance Frequency Analysis (RFA). The scale in Herz is non-intuitive and hard to communicate so it was translated to a scale of ISQ 1-99, more than 20 years ago. Today we know that the scale correlates to micro mobility and that high stability means > 70 ISQ, between 60-69 is medium stability and < 60 ISQ is considered as low stability.
ISQ, or Implant Stability Quotient, is a scale from 1 to 99 and is a measure of the stability of an implant. The ISQ is a close to linear mapping of the resonance frequency of the SmartPeg that is attached to the implant.
Most users agree, which is in line with the scientific studies, that a preferable second ISQ value for non-splinted implants should be ISQ 70 or more to initiate the prosthetic rehabilitation. For splinted implants which will stabilize each other, lower ISQ values could be used. More information about how to use the ISQ scale can be found under “Education”.
Osstell offers two types of SmartPegs: a single-use version and a reusable version. Each type has its own instructions.
Single-use SmartPegs
Single-use SmartPegs may be used for one or multiple implants during the same patient session but must not be reused at a later visit or for another patient.
They are not designed to withstand cleaning or sterilisation. The magnet cannot be autoclaved, and the aluminium threads are unprotected. Reusing a single use SmartPeg may result in inaccurate ISQ readings.
Reusable SmartPegs
Reusable SmartPegs can be used across multiple patient sessions, provided they are cleaned, maintained, and inspected according to the Instructions for Use. They can be reprocessed up to 20 cycles.
It is important to regularly check the SmartPeg for wear, thread deformation, or contamination. A damaged or worn reusable SmartPeg may affect measurement accuracy.
It is impossible to base the decision about immediate loading only on the ISQ-value. The literature shows that good primary stability is essential for a successful outcome, but there are of course also other parameters that have to be considered in such a decision. See also above about which ISQ values to expect. Many direct loading studies show good results with immediate loading if the primary stability is good. It seems as the implant stability will not change considerably from implantation to after osseointegration in many such cases.
The SmartPeg measures stability in all directions and singles out the most stable and the least stable directions. The two ISQ values represent these values. If they are both found at the same time, and they differ by more than 3 ISQ units, they are presented on the display together. The two values can also be found by measuring from two different directions.
There are currently thousands of articles where Osstell was used in the study, published in scientific papers. These abstracts could be found in our searchable database under “Resources/Scientific database“.
ISQ should always be measured in two directions, because implant stability can vary depending on the amount of surrounding bone in different directions. Measuring from two angles ensures that both the most and the least stable directions are captured.
To do this, take one measurement in the buccal-lingual direction and one in the mesial-distal direction. These two ISQ values represent the different stability directions. Sometimes the values are identical; sometimes they differ.
If the two measurements differ, always rely on the lowest ISQ value, as it reflects the weakest direction and therefore provides the most clinically relevant information about implant stability.
If you see an increased ISQ value, compared to the baseline value taken at placement, it is the
effect of osseointegration. The same is true for high values that should stay high over time. If there is
a significant drop in ISQ value, it is probably due to a failing implant.
Osstell helps you ensure that the implant is stable enough to load before initiating the prosthetic rehabilitation. This is especially important if you are trying to reduce treatment times or if you are treating risk patients.
Osstell measures the lateral stability of the implant and thereby indirectly the degree of osseointegration. The results are presented as an ISQ, ranging from 1-99 ISQ, which correlates to micro mobility.
Osstell measures implant stability using Resonance Frequency Analysis (RFA). The small SmartPeg that you attach to the implant has a tiny magnet on top. When the probe sends magnetic pulses, the SmartPeg starts to vibrate – similar to a very small tuning fork.
How it works:
- The probe sends magnetic pulses
These pulses make the SmartPeg vibrate without touching it. - The vibration changes depending on how stable the implant is
– A stable implant (good bone support) makes the SmartPeg vibrate faster.
– A less stable implant makes it vibrate slower. - The device converts this vibration into an ISQ value
– Higher stability → higher frequency → higher ISQ
– Lower stability → lower frequency → lower ISQ
It takes less than a second, and the ISQ value is shown immediately.
Periotests are designed for natural teeth that are attached to the bone with a periodontium. An implant is attached directly to the bone without periodontium.
Osstell uses a more sensitive scale with a non-contact, non-invasive technique which is totally objective and repeatable.
Torque is a one time, static measurement at placement of the implant and cannot be repeated later on in a non-invasive way. It measures the rotational friction between the implant surface and the bone and not the lateral stability of the implant. Osstell measures the lateral stability of the implant and the measurements could be repeated in a non-invasive, dynamic way to monitor the development of osseointegration.
You can have high percentage of bone contact in soft bone but low implant stability compared with a situation with dense bone, less contact percentage but still higher stability. This is explained by the stiffness in the bone itself (Young’s Modulus). In soft bone you will get a low ISQ at placement and you will see an increased stability value due to osseointegration. If you have high initial mechanical stability already at placement, osseointegration will not add stability in a significant way since the implant was already stable. If the implant does not osseointegrate the ISQ value will continue to decrease.
Yes. The ISQ value can be affected if the SmartPeg is not mounted correctly.
For accurate measurements, the SmartPeg should be attached finger tight using the SmartPeg Mount, which corresponds to about 4–6 Ncm.
At this level, the SmartPeg has firm, stable contact with the implant, and the resulting ISQ value is consistent – typically within ±2 ISQ units.
- If the SmartPeg is not fully seated:
The contact becomes unstable, and the ISQ value can appear artificially low, or you don’t receive any value. - If too much force is applied:
Overtightening does not improve accuracy. It may damage the SmartPeg threads.
– A reusable SmartPeg may be damaged so it cannot be used again.
– A single use or reusable SmartPeg may have its threads deformed, which can lead to inaccurate measurements during that session or, in worst case a broken SmartPeg.
In short: finger tight (4–5 Ncm) is enough to ensure stable contact and reliable ISQ readings.
If no ISQ value appears, it usually means the SmartPeg’s vibration is not being detected clearly by the instrument. Several factors can cause this, and most are easy to resolve:
- Distance to instrument is too great
The instrument should normally be held 2–4 mm from the magnet on top of the SmartPeg. Do not touch the magnet. If no value appears, move the instrument closer until the signal is detected. - Angle needs adjustment
The magnetic field is directional, so the orientation of the instrument matters. Measurements should be taken at approximately a 45° angle relative to the top of the SmartPeg. - Ensure you have the correct SmartPeg Type for the specific implant
Use this link to find the correct SmartPeg Type: Find my SmartPeg - Soft tissue damping
Surrounding tissue can sometimes dampen the vibration more than expected. Holding the instrument a bit closer usually compensates for this. - The implant platform is not clean
Ensure the implant platform is clean and free of debris, so the SmartPeg seats properly and transmits its vibration. - The SmartPeg is worn or damaged
• If you are using a reusable SmartPeg, make sure it has been inspected before use and that it is within 20 reprocessing cycles. A reusable SmartPeg that is worn out, bent, or has damaged threads will not vibrate correctly — and the instrument may not be able to pick up a reading.
• A single‑use SmartPeg is intended for one treatment session only.
• Ensure the SmartPeg is properly seated on the implant and that it is not in contact with surrounding tissue, as this can prevent accurate signal detection and may result in no ISQ value or an incorrect one. - Electromagnetic interference
Strong electromagnetic noise sources nearby may disturb the measurement. (See the FAQ on “Electromagnetic noise” for details.)

The average ISQ is approximately 70. The clinical range that can be seen is from 40-45 up to 85-90. Most implants have between 55 ISQ to 80 ISQ
Clinically, the lowest number seen is approximately 40 ISQ. If the stability is less than 30, the implant is more or less “loose”. Values below 20 will not be seen, unless on a lab model with artificially low stability.
Yes, you can measure ISQ on a patient who has a pacemaker. But to be really sure, you should keep in mind to position the probe away from the chest/pacemaker due to the magnetic pulses emitted. There are numerous reports about pacemakers and magnetic fields, e.g. this report.
Measure at implant placement and again before the final restoration to monitor the development of osseointegration. If the ISQ value is not high enough it is advised to add time and remeasure before proceeding with the prosthetic rehabilitation of the implant.
